Plant genome research has laid the foundation for discovery of genes and networks underlying economically important traits in crop plants. However, the ultimate goal to link genotype to phenotype remains difficult, and one of the greatest challenges is identification and characterization of sequence variants underlying quantitative variation. A major limitation for application of genome data to improve crops is that knowledge of genomic diversity, specifically structural variations (SV), is incomplete, despite several examples of significant roles in agriculture. This project will focus on dissecting SVs in tomato and its ancestors, to elucidate how SVs shape plant genomes and contribute to domestication and improvement. The project will therefore develop a comprehensive genome-level view of SVs to aid in linking them to complex agricultural phenotypes. A second related goal is to test the hypothesis that known domestication and improvement genes and variants can be exploited to engineer domestication. To achieve this, gene editing, with its power to create SVs, will be used to create new crops from the stress-tolerant progenitor of tomato and a distant Solanaceae relative that is an emerging fruit crop.
Plant genome research has laid the foundation for discovery of genes and networks underlying economically important traits in crop plants. However, the ultimate goal to link genotype to phenotype remains difficult, and one of the greatest challenges is identification and characterization of sequence variants underlying quantitative variation. A major limitation for application of genome data to improve crops is that knowledge of genomic diversity, specifically structural variations (SV), is incomplete, despite several examples of significant roles in agriculture. This project will focus on dissecting SVs in tomato and its ancestors, to elucidate how SVs shape plant genomes and contribute to domestication and improvement. The project will therefore develop a comprehensive genome-level view of SVs to aid in linking them to complex agricultural phenotypes. A second related goal is to test the hypothesis that known domestication and improvement genes and variants can be exploited to engineer domestication. To achieve this, gene editing, with its power to create SVs, will be used to create new crops from the stress-tolerant progenitor of tomato and a distant Solanaceae relative that is an emerging fruit crop.
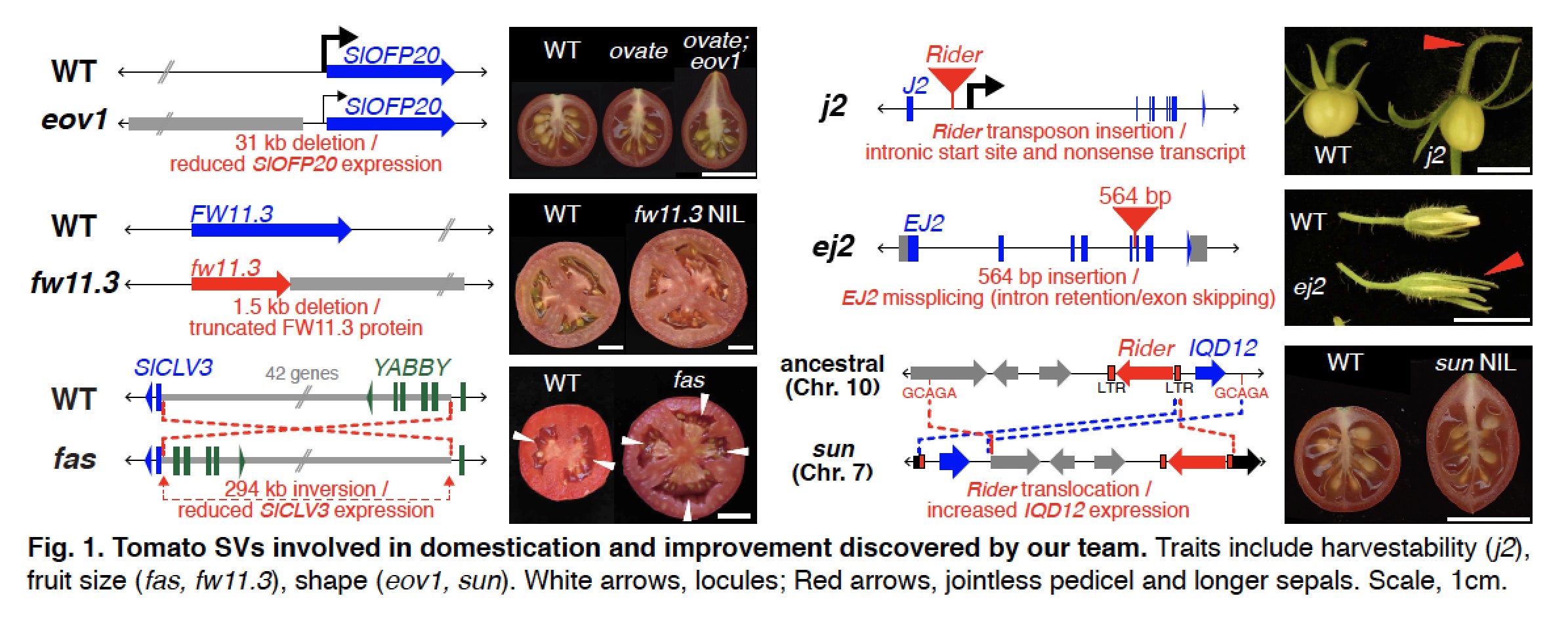
Limited knowledge on the extent and diversity of structural variation in plant genomes is hindering the ability to link genes to important crop phenotypes. This project will unite new long-read sequencing technologies, computational biology, developmental and quantitative genetics, and genome editing to elucidate and manipulate structural variation (SV) at a scale never before achieved for a major crop. Tomato provides a powerful system due to its relatively small and high quality reference genome and availability of resequenced genomes. By applying SV-detection algorithms to existing short-read Illumina sequencing data from hundreds of accessions, more than 40 genomes will be selected, capturing the majority of predicted SV diversity, to establish new reference genomes using the latest long-read sequencing technology (PacBio and 10X Genomics). From these data, a compendium of validated SVs will be generated and integrated with ongoing genome-wide association studies. Significant gene-associated SVs, including those affecting gene activity measured by genome-wide transcript profiling, will be characterized using CRISPR/Cas9 gene editing and quantitative phenotypic analyses, focusing on reproductive traits that drive crop productivity. In parallel, CRISPR/Cas9 gene editing will be used to generate a collection of SV mutations in known yield and fruit quality genes in two related wild Solanaceae with agricultural potential, with the goal of achieving major steps towards domestication and for comparative developmental genetics studies. This project will greatly expand our knowledge of genomic diversity in tomato, and provide a road map for dissecting SVs in other crops, where such knowledge can be exploited to improve productivity.
In addition to the scientific advances and the potential to bring significant improvements to US agriculture, this project will train young scientists at multiple levels, as well as promote outreach and education in plant biology. Project personnel will expand an existing educational program to highlight the importance of plant development, genomics, and gene editing technology in crop improvement, and will present these concepts in inner-city schools in NY. Hands-on experiments with plants and molecular biology will be used to educate elementary and middle school students, parents and teachers about the importance of integrated disciplines in plant research to improve agriculture.
Project meeting at Cold Spring Harbor Laboratory, July 2019